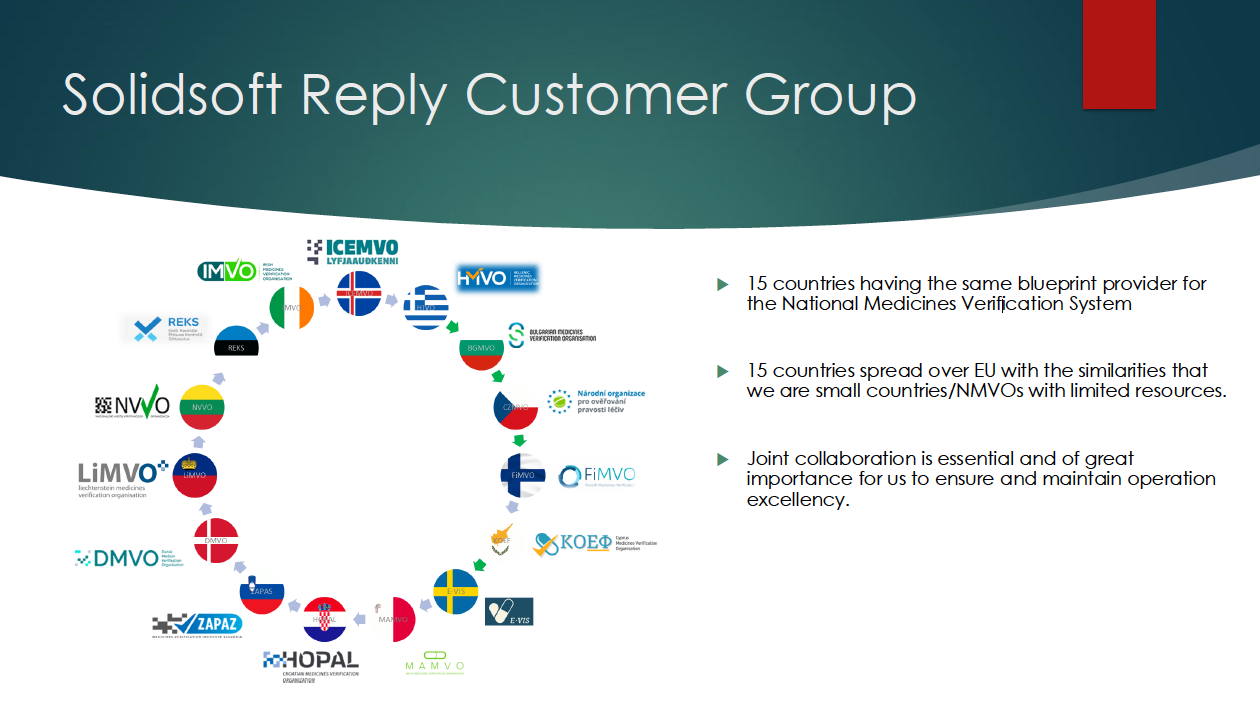
The group of countries with NMVS from Solidsoft Reply is growing
Medicines verification in the context of Falsified Medicines Directive (FMD), so far, have been managed in the national medicines verification systems (NMVS) provided by 2 suppliers. Individual countries decided which supplier is prefered by them. One of them is British company Solidsoft Reply, whose system is also operated in the Czech Republic by Czech Medicines Verification Organization (CZMVO). The group of 12 countries that (like CZMVO) have been using the Solidsoft Reply system, expanded in 2024 - Finland changed the supplier. The NMVS was successfully launched in Greece in February 2025; Solidosoft Reply (SSR) is also a supplier.
Estonia is now preparing to change from Arvato, and the system will be migrated to a production environment on April 27, 2025.
The group of countries with the same so-called blueprint NMVS supplier - Solidsoft Reply - now has 15 members. All the national organizations of these countries, now including the Estonian REKS, cooperate at different levels within the customer group in the EMVS, sharing resources and knowledge not only in testing, quality, auditing and change management. The SSR country group acts together towards EMVO and other stakeholders to ensure that not only costs, but also the whole EMVS system is optimized to comply with the Commission Delegated Regulation (EU) 2016/161.

 Registration and login to systems
Registration and login to systems












